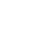
ESSENCE Platform
The ESSENCE chip represents an advanced non-planar electrochemical sensor technology capable of multifaceted applications, ranging from detecting emerging contaminants like PFAS to addressing the challenges of zoonotic diseases. This modular, user-friendly, and portable sensor chip is highly adaptable, making it a versatile tool for the rapid detection of a wide array of emerging contaminants and biothreats.
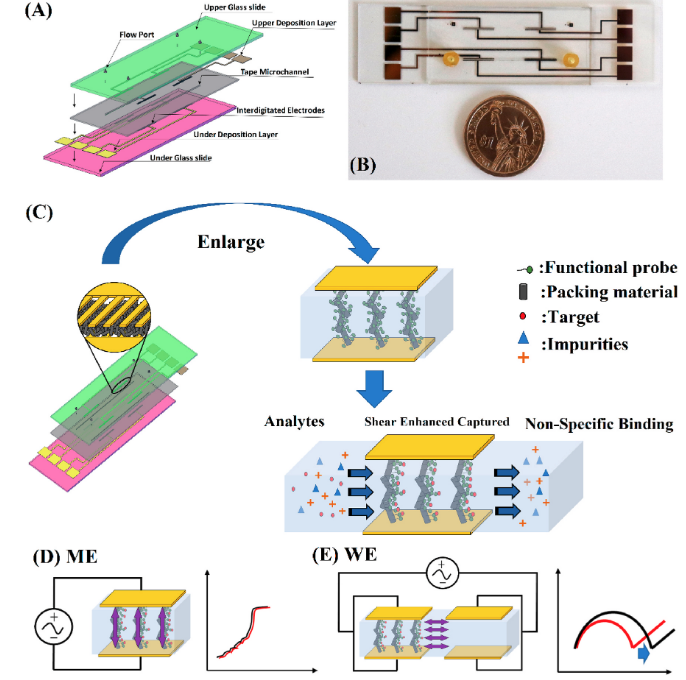
PFAS Sensor
PFAS, or per- and polyfluoroalkyl substances, are a group of synthetic chemicals that have gained significant attention due to their widespread presence and potential environmental and health risks. These persistent compounds are known for their water and grease-resistant properties. They have been used in various consumer and industrial products, including non-stick cookware, firefighting foams, and stain-resistant fabrics. Concerns arise from their persistence in the environment, bioaccumulation in organisms, and potential health effects, which include links to various adverse health outcomes such as cancer, reproductive issues, and developmental problems. As a result, there is ongoing research and regulatory efforts to mitigate their impact and limit their use in various applications. At the Basuray lab, we work on a microfluidic electrochemical sensor (µFES) to detect PFAS in water samples. Using a Metal-Organic Framework (MOF) based transducer and packing material, we have developed a preliminary version of the ESSENCE platform for detecting PFOS. Current work is modifying the PFAS detection platform to improve reliability and sensitivity.
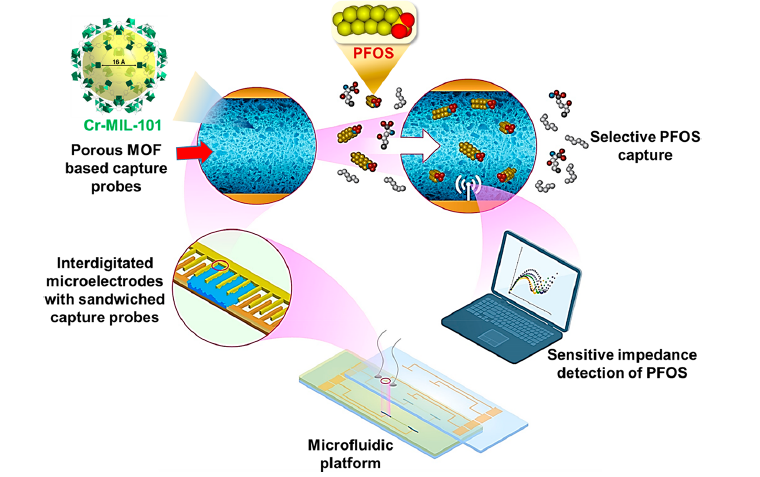
Heavy metal ion detection
Over the past few years, there has been a growing global concern about the impact of heavy metal (Cu2+, Hg2+, Pb2+, Cd2+, and others) contamination on the environment and public health. This is because these metals are used more frequently in various industrial, agricultural, domestic, and technological applications, significantly increasing human exposure. Drinking water contaminated with heavy metals is the primary source of human exposure to such toxins. One of the projects in our lab is heavy metal detection using microfluidic electrochemical sensors (μFESs). Finite element analysis using COMSOL Multiphysics is carried out to optimize the electrode structure of μFESs for sensitive ion detection. Two devices with different electrode structures (planar interdigitated and non-planar interdigitated) have been prepared for heavy metal detections.
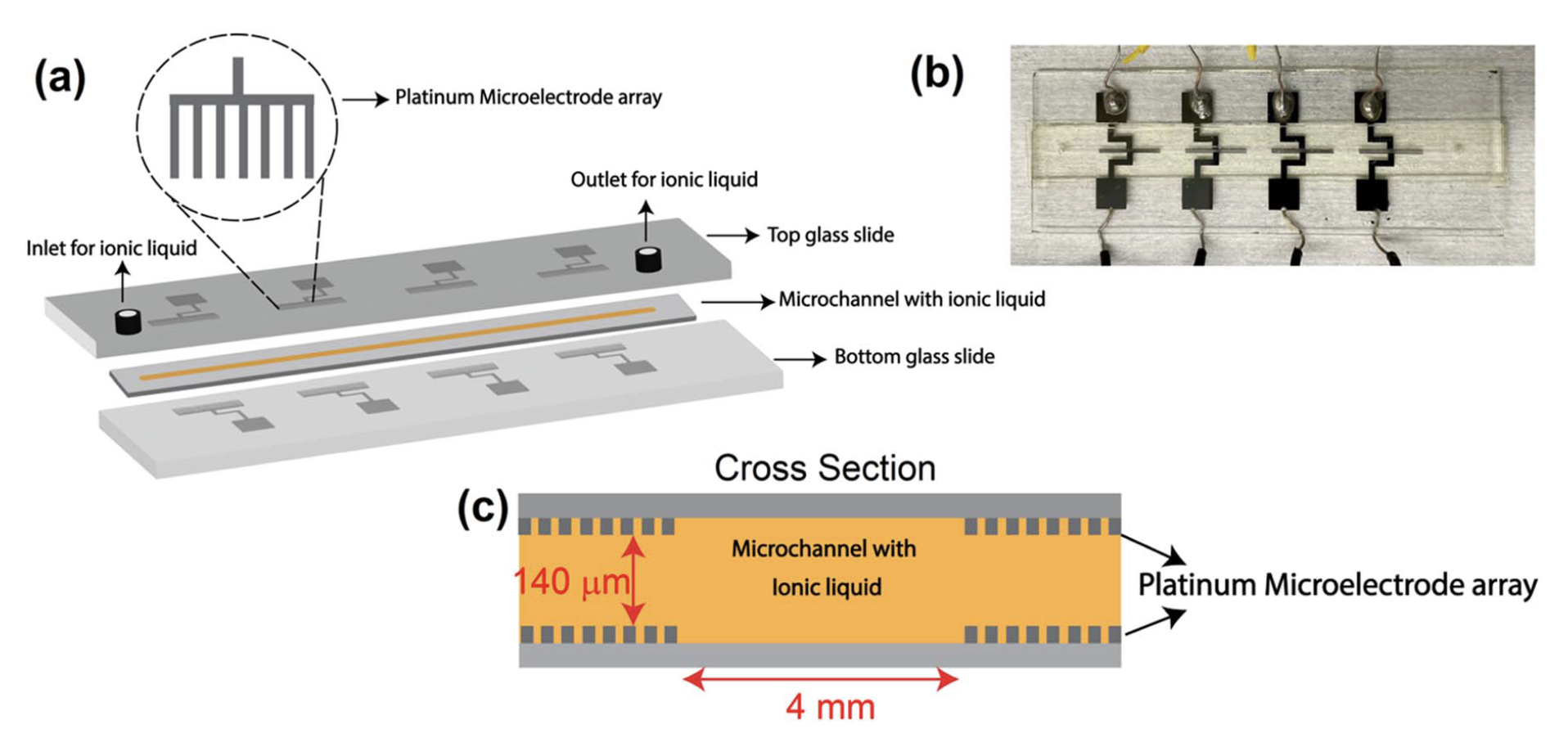
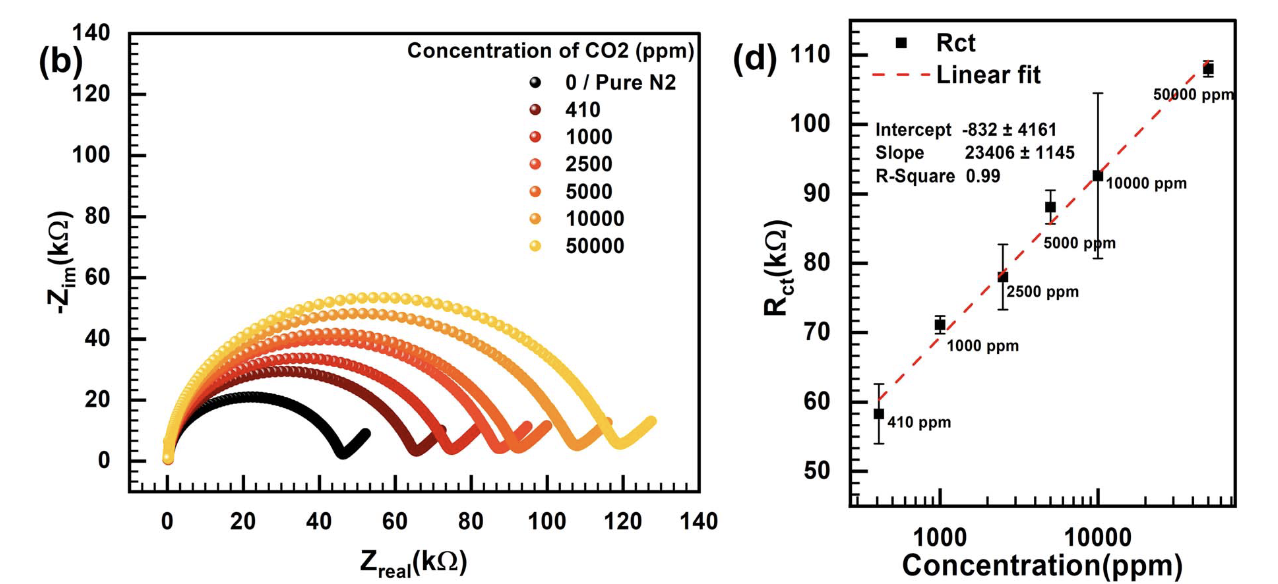
Detection of volatile organic compound
Detection of volatile organic compounds (VOCs) is of great importance in fields like breath diagnostics, antibiotic susceptibility tests, and early plant disease diagnostics. However, the current sensors face two main limitations: 1) Low sensitivity and 2) poor selectivity. The limit of detection for VOC sensors needs to be in ppb or at a low ppm level to be used in the applications stated above. The VOC sensor should also be able to distinguish between different VOCs even of the same class (alcohols, ketones, and alkanes) to be useful in the above applications. To achieve this goal, our lab is developing a microfluidic electrochemical gas sensor. We believe that combining microfluidic architecture with an electrochemical detection technique can significantly enhance the sensitivity of the sensor. The microfluidic architecture also allows the use of a very low volume of sensing material/electrolyte thereby reducing the cost of the sensor and allowing the sampling of small amounts of gases. To enhance the selectivity of the sensor in detecting multiple VOCs, multiple electrochemical detection techniques like EIS, CV, DPV, IV, and amperometry can be used in tandem to gain as much information and use ML and AI techniques to confidently differentiate between different VOCs. Currently, for this project, our lab is developing and optimizing sensor architectures using carbon dioxide as the model gas and ionic liquid as the sensing material.